Last week we sampled for radiocarbon from the winter warming and control sides of the snow fence experiment.

What is radiocarbon?
Radiocarbon dating or “radiocarbon” as we call it for short, is a technique used to determine the age of carbon. It has been used to date mummies excavated in Egypt, an [iceman] (http://news.discovery.com/history/archaeology/iceman-discovery-110919.htm) found in Italy, and, you guessed it, the age of carbon in the permafrost.
How does radiocarbon work?
Carbon has three main isotopes: C12, C13, and C14. C14 is formed in the atmosphere through a reaction between nitrogen and cosmic rays. Unlike C12 and C13, C14 is unstable and decays back to nitrogen over time. The half-life of this decay process is around 5,700 years. So, if you have a pile of C14 and left it for 5,700 years, when you (or your descendants returned), you would only have half of that pile left.
All living things take up C12 and C14 (through photosynthesis or respiration) in the same ratio that is in the atmosphere. So when an organism dies, the ratio of C12 and C14 in the atmosphere is preserved in the body of the organism. Over time, the C14 in the dead organism (in our case, mostly dead plant material) decays but the C12 stays constant. By comparing the ratio of C14 to C12 in this dead organism (which will be less than when it died) to the ratio of a living organism, we can determine how long it has been dead (or, the age of the C).
Why do we care about the age of the C in the permafrost?
Previous work with radiocarbon from Healy has showed that areas with more permafrost thaw are releasing older C:
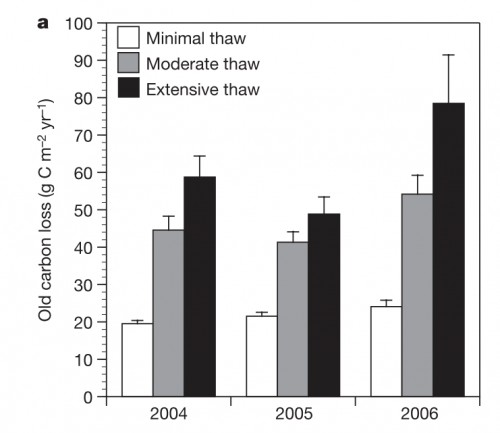
This has important implications for feedbacks to climate change. The fact that areas with increased thaw are releasing older C means that plants and microbes may be starting to respire C that has been stored in the permafrost for millennia. This means we may be starting to chip away at the huge amount of C stored in permafrost soils (because the newer C has already been respired). This C, of course, will be released to the atmosphere in the form of a green house gas and further warm the climate.
I sampled radiocarbon from the winter warming and control plots. This is to see if warming causes the release of older C during the winter.
How do we sample radiocarbon?
We run air from previously installed chambers through a trapping system:

which then feeds the air into a molecular sieve:

The molecular sieve captures all the C. We then cap the sieve as fast as possible so we don’t get any atmospheric air contamination:

After we sampled from all plots, we send the sieves to our lab manager (Grace Crummer) in Florida. Grace then heats the sieves to 625°C to release the C for radiocarbon analysis.



Comments