Our Arctic core samples keep turning up great surprises! If you'll recall from a previous journal (August 13, 2010 Piston Coring), we struck gas hydrate in the bottom of one of our cores in the Beaufort SeaThe Beaufort Sea lies to the north of Alaska and the Yukon and Northwest Territories.. At the core analysis workshop at the USGS Coastal & Marine Science Labs in Menlo Park, CA earlier this month we discovered that some of the core sections from that site also had numerous clam shells. Since each core is only about 8 cm wide, that would mean that this part of the sea floor is pretty dense with clams! Many were intact with both top and bottom valves (shells) in place indicating they lived there instead of being carried there later. But what would clams live on in this cold, dark, deep water??

As you probably know, most ecosystems on Earth rely on sunlight. Photosynthetic organisms (such as plants or algae) convert light into stored energy in the form of carbohydrates. Other organisms may consume these producers, and other organisms may eat those, etc. all the way up the food chain. Indeed, typical clams filter feed plankton from sea water and would quickly starve if the Sun stopped shining. Until fairly recently you could still find biology textbooks stating that all ecosystems on Earth depend on sunlight. It turns out that this isn't true, though. There is another type of ecosystem on Earth known as a chemosynthetic community. In these ecosystems the base of the food chain consists of bacteria that store energy by utilizing chemicals such as hydrogen sulfide or methane. Other organisms can take advantage of these bacteria through symbiotic relationships, providing a secure place for the bacteria to live and delivering the needed chemicals to the bacteria while "harvesting" food energy from them. The submersible Alvin famously discovered diverse chemosynthetic communities at mid-ocean ridge hydrothermal (hot water) vents in the 1970s. More recently another type of marine chemosynthetic community known as a cold seep has been found that relies on the seepage of suitable chemicals upward through marine sediments in deep cold water. The combination of numerous clam shells, hydrogen sulfide odor, and evidence of methane in the core all point to our discovery of an Arctic cold seep community in the Beaufort SeaThe Beaufort Sea lies to the north of Alaska and the Yukon and Northwest Territories..
Cold seep clams can absorb and concentrate hydrogen sulfide (a chemical toxic to "normal" animals) and deliver it to chemosynthetic bacteria living in their bodies. The bacteria convert this into food that they and the clams use. For these types of organisms, it doesn't matter if the Sun rises tomorrow! This not only has important implications for life on Earth but shows us another significant type of habitat for possible life to exist in throughout our Solar System and beyond.
To get a handle on the sea-bed processes going on here, geochemist Tom Lorenson & biogeochemist John Pohlman collected several types of samples along the core sections for detailed analysis. Traces of methane left stuck to sediment grains (adsorbed gas) or trapped between sediment grains (free gas) will undergo isotopic analysis that can help to pinpoint its origin- for example, is it biogenic (produced by bacteria breaking down organic matter) or thermogenic (produced by heat & pressure acting on deeply buried organic material)? Similarly, carbon deposited in authogenic carbonate nodules and in the clam shells themselves will undergo isotopic analysis to delve into its origin. Pore water (the water between sediment grains) was collected at intervals to later undergo many isotopic and chemical tests. By taking these samples along the length of the core, they can also track the movement of cold seep chemicals up through the sediment as well as sea-water compounds down through the sediment and study the interaction of each in the mud.
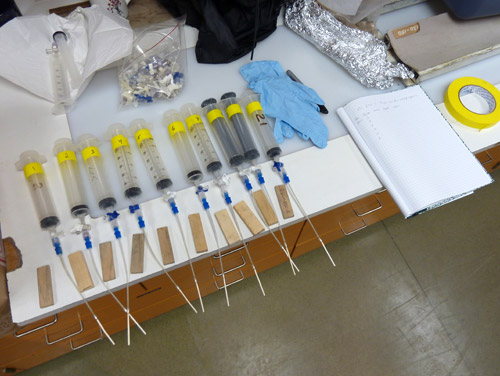
Getting water out of fine sediments can be tricky- it won't just drain out. One solution is to take pucks of sediment and squeeze the water out, but this requires more destructive sampling of the core and may introduce water from within clay grains rather than just pore water. Instead, John employed a neat technique called rhizon sampling. This technique relies on the capillary action of water- to see this in action just dip a corner of a paper towel into a glass of water and watch the liquid climb up into the paper towel against gravity! A rhizon tube has microporous material (about 1 micron mean pore width) inside which both filters the water from the sample and wicks it out through capillary action. A rhizon tube is inserted into the targeted strata and connected to a syringe to collect the sampled water. The syringe plunger is pulled back and held there with a wood block as it can take hours to collect a few milliliters of water this way. Our rhizons were left in place overnight in the walk-in core storage refrigerator in order to collect sufficient water for the battery of tests it will undergo.


Here's a video showing rhizon sampling in action! That's all for now- Bill http://


Comments